Aortic valve insufficiency is the leakage of clean blood back into the left ventricle instead of being pumped to the body because the aortic valve closes defectively. This structural problem is characterized by symptoms such as shortness of breath, rapid fatigue, and palpitations, especially during movement. The condition is classified as mild, moderate, and advanced degrees according to echocardiography measurements. Its treatment varies depending on the level of the disease and includes effective procedures ranging from medication-based follow-up to the sutureless TAVI method or surgical valve replacement. Timely correction of this mechanical leakage is vital to prevent the heart muscle from becoming exhausted.
What Is Aortic Valve Insufficiency and What Kind of Problem Does It Cause in Our Heart?
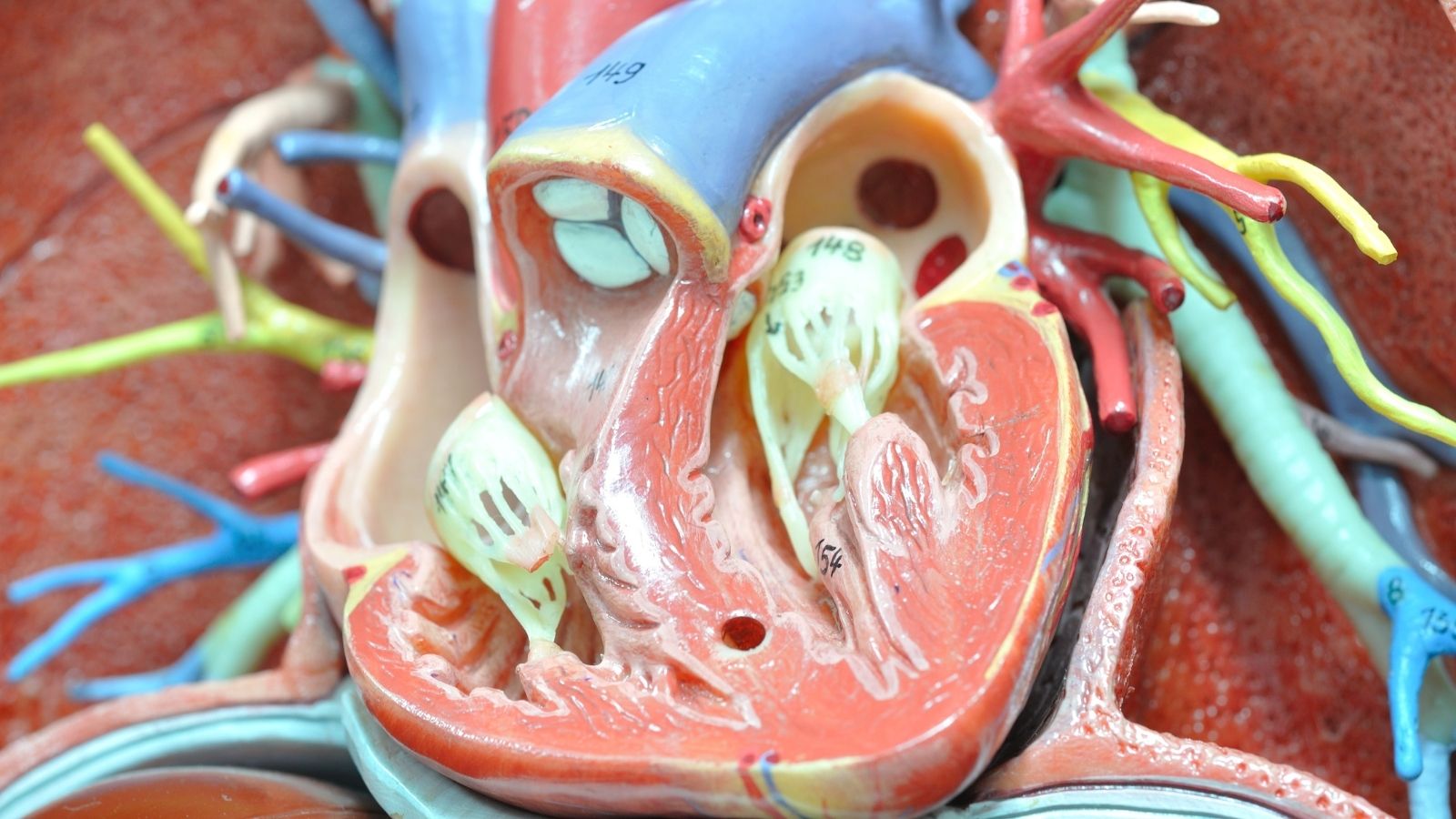
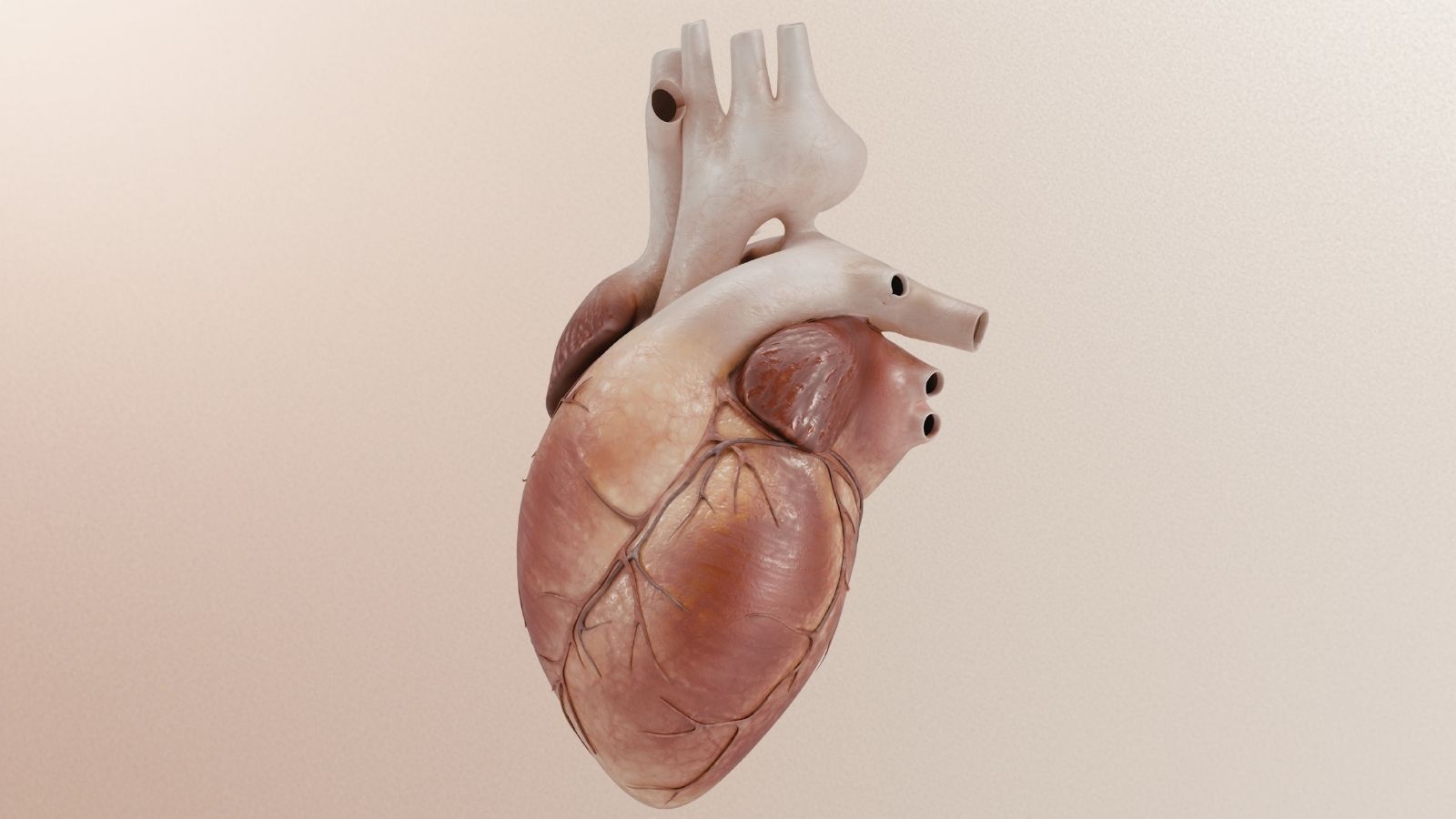
When the left ventricle, the lower left chamber of the heart, contracts, it ejects the clean blood inside it into the aorta with great pressure. At the moment of this ejection, the leaflets of the aortic valve open fully and allow the blood to pass easily. In the momentary resting phase, which lasts less than a second after the heart finishes contracting and starts relaxing to fill with new blood, the aortic valve must close suddenly and tightly. In this way, the blood that has been pushed into the vessel cannot return into the heart due to gravity or intravascular pressure and is forced to continue its path toward the body.
In aortic valve insufficiency, because the structure of the leaflets that form the valve is impaired, has lost its flexibility, or has moved away from each other, complete closure cannot be achieved. A gap, a leakage space, remains between the valve leaflets. The moment the heart relaxes, a considerable portion of the blood sent to the body begins to flow backward through this gap, that is, back into the heart. This situation leaves the left ventricle of the heart in the middle of a difficult mechanical problem. The ventricle has to receive fresh clean blood from the lungs on one hand, while also accepting the old blood leaking back from the aorta on the other. The volume of blood accumulated inside rises far above normal. In the next contraction, the heart has to spend much more power than before and contract much more strongly to eject both the blood it normally needs to pump and the excess blood leaking back at the same time. This endless extra workload gradually lays the groundwork for cellular deterioration that deeply affects the structure, shape, and working capacity of the heart muscle.
Does Aortic Valve Insufficiency Develop Suddenly or Progress Insidiously Over the Years?
The development of this structural problem and the damage it causes to the heart show dramatic differences depending on the underlying cause. The disease usually appears as a very insidious and slowly progressing chronic process that lasts for years, even decades. In this slowly progressing process, the heart activates a very intelligent adaptation mechanism to carry the increasing fluid load it is exposed to. To accommodate the increased blood volume, it enlarges its internal volume and thickens its muscle walls to preserve its ejection power. Just as the muscles of an athlete who regularly lifts weights grow over time, the heart also enlarges in response to the increased workload. This enlargement process allows the patient to live a very active life for many years without feeling any signs of disease. However, the heart muscle’s capacity to stretch and thicken is not unlimited. Once a certain threshold is exceeded, connective tissue increases between the heart muscle cells, elasticity is lost, and contraction power begins to weaken permanently.
On the other hand, in some unfortunate situations, the structure of the valve may deteriorate suddenly, within hours or days. Severe bacterial infections that directly attack the valve or sudden tears occurring in the aorta can suddenly render the valve nonfunctional. The heart cannot find the time it needs to prepare for this sudden and massive fluid flood, to stretch, or to strengthen its muscles. As blood far beyond its capacity suddenly fills inside, the pressure within the heart rises rapidly. This high pressure reflects backward and hits the lungs. The air sacs in the lungs rapidly fill with fluid, making it impossible for the patient to breathe. With the sudden drop in the amount of blood pumped to the body, blood pressure collapses and the nourishment of all organs is impaired.
What Are the Main Causes and Risk Factors That Lead to Aortic Valve Insufficiency?
Understanding the factors that disrupt the valve’s closing mechanism is vital for preventing and treating the disease. This problem may sometimes originate from diseases that directly affect the valve’s own tissue, and sometimes from deformation of the frame in which the valve sits over time.
The most common causes that lead to valve deterioration are as follows:
- Bicuspid aortic valve
- Rheumatic heart disease
- Infective endocarditis
- Aortic enlargement
- Aortic aneurysm
- Uncontrolled high blood pressure
- Marfan syndrome
- Calcification related to advanced age
A congenital structural difference that is quite common in society is one of the most frequent sources of the problem. Under normal conditions, the aortic valve consists of three thin leaflets that close perfectly against one another. However, in some individuals, this valve forms with two leaflets while developing in the womb. Although it functions normally at first, this two-leaflet structure is more vulnerable to the dynamics of blood flow. Exposed to unbalanced stress with every heartbeat over the years, the valve eventually begins both to thicken and calcify and to leak by sagging from its edges.
Febrile illnesses caused by untreated throat infections during childhood may also make the heart pay the price in later years. Antibodies produced by the body to fight infection mistakenly attack the heart valves, causing the leaflets to thicken, shrink, and fuse with one another. Apart from this, bacteria that enter the bloodstream and directly attach to the heart valve, multiply there, and destroy the tissue can also cause serious leaks.
Sometimes, even if the valve leaflets are completely healthy, the root part of the aorta where the valve sits becomes diseased. High blood pressure left untreated for years applies continuous pressure to the vessel walls and gradually widens the diameter of the vessel. As the vessel widens, the valve leaflets attached to it move away from each other. Since the leaflets cannot meet tip to tip, a wide opening remains in the middle and blood leaks backward through this opening.
What Kind of Symptoms Does a Person with Aortic Valve Insufficiency Feel in Their Heart in Daily Life?
During the silent and insidious period of the disease that lasts for years, patients generally feel completely healthy. They can exercise, work in physically demanding jobs, and continue their daily lives without any restriction. The heart’s ability to adapt is so successful that it does not reflect the internal wear outward. However, once the critical threshold where the heart muscle begins to tire and give up is crossed, the body’s alarm bells begin to ring. Complaints usually emerge not when the patient is at rest, but during exertion, when the heart has to pump more blood.
In the period when the disease becomes clinically symptomatic, the most common complaints experienced by patients are as follows:
- Shortness of breath
- Rapid fatigue
- Palpitations
- Chest pain
- Dizziness
- Swelling in the ankles
- Feeling of pulsation in the neck
Patients begin to feel the need to stop and rest frequently on slopes or stairs that they could previously climb very easily. A feeling of pressure, as if there is a heavy load on the chest, causes exercise capacity to gradually decrease over months. Waking up suddenly at night with shortness of breath and a choking sensation while lying flat is a very important sign that the heart has begun to accumulate fluid backward in the lungs.
In addition, because the amount of blood the heart ejects with each contraction is the sum of both the blood pumped forward and the blood leaking backward, the pulse wave formed in the vessels is very strong. Patients may feel their heartbeats as powerful pounding sensations, especially in quiet environments while lying down, in their neck, ears, and even abdomen. When the blood flow going to the heart’s own vessels decreases due to an excessive drop in diastolic blood pressure, the person may experience tightness and burning-type pain in the chest during exertion.
Which Methods Are Used to Make the Definitive Diagnosis of Aortic Valve Insufficiency When It Is Suspected?
Even a simple physical examination can provide very strong clues about the disease. When the chest is listened to with a stethoscope, that typical blowing-like sound caused by blood leaking backward can be easily noticed. In blood pressure measurements, a very widened difference between systolic and diastolic blood pressure is also an indicator of the hemodynamic imbalance created by this leakage. However, advanced imaging technologies are needed to make a definitive diagnosis, determine the degree of the disease, and measure the extent of the damage it has caused to the heart.
At this point, the most basic and most valuable diagnostic tool is echocardiography. This method, which uses sound waves to display the internal structure of the heart, its chambers, and the movements of the valves live on the screen, is a completely painless and radiation-free procedure that does not cause any discomfort to the patient. Thanks to advanced color mapping techniques, the thickness and length of the blood jet spraying backward through the gap where the valve does not close completely, and the volume of blood leaking backward in milliliters during one heartbeat, are calculated very precisely. Sometimes, when more detailed images are needed, the heart can be examined much more clearly from behind with a camera at the tip of a thin tube lowered through the esophagus, just like endoscopy. When necessary, cardiac MRI scans or tomographies also map the diameter of the aorta and the structure of the valve with millimetric precision.
Which Measurements of the Heart Are Taken into Account When Deciding the Timing of Aortic Valve Insufficiency Treatment?
When the degree of the disease reaches an advanced level and the patient begins to experience complaints such as shortness of breath and fatigue, making the treatment decision is quite clear. Because replacing or repairing the valve has now become necessary; no medication treatment can restore a torn or stretched valve to its former state. Medications only temporarily relieve the patient by reducing fluid accumulation or balancing blood pressure, but they do not solve the underlying mechanical problem. The truly critical decision is made in patients who have no complaints yet but have advanced leakage. In these patients, intervening on the valve too early means taking an unnecessary procedural risk, while being too late causes irreversible damage to the heart muscle.
To establish this delicate balance and determine the correct timing of intervention, some critical limits are observed during follow-up performed with echocardiography.
The critical measurement parameters closely monitored are as follows:
- Ejection fraction
- End-systolic diameter
- End-diastolic diameter
- Regurgitant volume
- Vena contracta width
When the diameter of the heart during contraction exceeds a certain millimeter value, this shows that the heart muscle has now stretched excessively and is beginning to lose its ability to recover. Likewise, when the ejection fraction, which is the percentage of blood inside the heart that is ejected to the body, starts to fall below normal limits, alarm bells ring. If these critical limits are exceeded, even if the patient feels very well, intervention on the valve is essential to prevent permanent heart failure that may develop in the future.
Which Nonsurgical Treatment Methods Stand Out Today in the Treatment of Aortic Valve Insufficiency Apart from Traditional Open-Heart Surgery?
Until the recent past, the only and most definitive solution for serious structural deterioration of the aortic valve was classical open-heart surgery. This well-established method involves cutting the chest cavity open from the middle, completely stopping the heart, transferring blood circulation in the body to an artificial heart-lung machine during the procedure, then cutting out the damaged valve with a scalpel and suturing a new biological or metallic valve in its place. Undoubtedly, this method is extremely reliable, permanent, and a standard that medicine has successfully applied for many years. However, the long intensive care periods after the procedure, the months-long restrictions required for the breastbone to heal, and the risks arising from the nature of the operation have led both patients and physicians to seek less exhausting alternatives.
Thanks to the dizzying point reached by medical technology today, the TAVI procedure offered by interventional cardiology, that is, replacement of the valve by the catheter method, has opened a brand-new chapter in the treatment of valve diseases. Initially developed only for elderly patients or those considered too risky to tolerate open surgery, this method is now being successfully applied to an increasingly wider patient population as its success has been proven. In this method, the patient’s chest cavity is definitely not opened, the heart is not stopped, and in most cases there is not even a need to put the patient completely to sleep, that is, to use general anesthesia.
How Is the TAVI Procedure Performed Step by Step Through the Groin in Patients with Aortic Valve Insufficiency?
The patient is taken to an angiography laboratory specially designed for the procedure and equipped with advanced imaging devices. Only the groin area is numbed with local anesthesia, and mild sedation is given so that the patient feels comfortable during the procedure. Through a small needle hole, the main artery in the groin area is entered, and thin, flexible guidewires are placed inside the vessel.
The biological heart valve, previously selected according to the patient’s vascular and cardiac anatomy, is tightly folded and compressed like an umbrella in a special laboratory environment and enclosed inside a delivery sheath. This sheath is inserted into the vessel through the hole in the groin and slowly advanced toward the heart along the body’s main vascular route. Thanks to the high-resolution X-ray images on the screen, every millimetric movement of the valve is followed instantly from outside. The new valve is positioned exactly inside the old and leaking valve in the heart, with its alignment adjusted millimetrically.
The moment the valve opens is the most critical seconds of the procedure. To prevent the new valve from being dragged by blood flow or shifting from its place while opening, very rapid impulses are sent to the heart with the help of a temporary pacemaker. The heart rate is momentarily increased up to around two hundred beats per minute; in this way, the heart trembles in a contracted state, but because it cannot fill with blood and eject it, blood flow comes almost to a halt for a few seconds. During exactly these stormless seconds, the delivery sheath is withdrawn or the balloon inside it is inflated. The compressed new valve opens strongly within seconds, takes its original form, and grips tightly onto the walls of the old valve. The moment the heart rate returns to normal, the new valve opens and closes perfectly, immediately stopping the leakage. At the end of the procedure, the small entry hole in the groin is closed from the inside with special systems.
Can TAVI Also Be Performed in Pure Aortic Valve Insufficiency Without Valve Calcification or in Patients Who Have Previously Had Open Surgery?
In the early years of the TAVI procedure, the technology needed hard and calcified tissues in the vessel walls for the valve to hold firmly. Since calcification was abundant in patients with aortic valve stenosis, the prosthetic valve could catch onto this calcium and remain fixed. However, in pure aortic insufficiency cases where there is no calcification and the leaflets only stretch and sag, the risk of the new valve slipping out of place was very high because there was no protrusion for it to hold onto. For this reason, patients with pure insufficiency were deprived of this technology for a long time. However, today’s engineering marvel new-generation valve designs have succeeded in solving this problem as well. These new systems, which have special clamping mechanisms on them, hold the patient’s own loosened leaflets like a paper clip and allow the prosthesis to lock into the heart. Thus, even without calcification, the valve sits securely in place without slipping.
In addition, technology offers a great miracle for patients who previously underwent open-heart surgery and received a biological valve, but whose valve has degenerated and started leaking again over the years. In the past, these patients would have needed a second open surgery, which was much riskier, to replace the degenerated valve. Now, with a method called valve-in-valve, a new TAVI valve can be placed through the groin exactly in the center of the old valve without trying to remove it at all. The old valve serves as a perfect housing for the new valve.
What Should Patients Pay Attention to in Their Daily Lives During the Recovery Process After Aortic Valve Insufficiency Intervention?
The greatest comfort provided by interventional methods is the rapid and painless recovery process after the procedure. Only a few hours after the TAVI procedure performed through the groin, patients can sit up in bed and eat, and the next day they can walk in the corridor. When everything goes well, they are generally discharged within two or three days and return to their own homes and familiar beds. Since the chest cavity is not cut, postoperative pain, infection risk, and long-term movement restrictions are not experienced. Patients can return to driving, daily tasks, and social life in a very short time. However, this rapid recovery should not mean that patients leave themselves completely uncontrolled.
The main rules to pay attention to during the recovery process are as follows:
- Not skipping medications
- Not straining the groin
- Not lifting heavy objects
- Complying with doctor checkups
- Informing the dentist
To support the healthy functioning of the newly implanted valve, it is very important to strictly follow the timing of blood thinners or blood pressure-balancing medications prescribed by the physician. To allow the groin area where the procedure was performed to heal, hard movements that excessively stretch that area and carrying heavy loads should be avoided in the first weeks. Perhaps one of the most important issues to pay attention to is the risk of infection. Oral and dental health is directly related to the heart. Before any tooth extraction, root canal treatment, or bloody surgical procedure, the patient’s informing the relevant physician that they have undergone heart valve surgery and using preventive antibiotics before the procedure is of vital importance to prevent bacteria from settling on the valve.
Which Foods Should Patients with Aortic Valve Insufficiency Avoid to Protect Their Heart?
In this disease picture, where the heart is already struggling with an increased blood volume and pressure, the patient’s eating habits will either lighten the burden on the heart or make its job more difficult by tiring it further. Permanent changes in the diet are essential to protect the structure of blood vessels, prevent the amount of fluid inside the vessels from getting out of control, and prevent high blood pressure attacks. In particular, foods that cause fluid retention in the body and vascular stiffness must definitely be avoided.
Some of the foods that should be avoided or whose consumption should be limited are as follows:
- Table salt
- Ready-made pickles
- Processed sausages
- Margarine
- Tail fat
- Fried foods
- Sugary drinks
- Energy drinks
Salt, which is at the top of this list, is the biggest insidious enemy of patients with aortic insufficiency. High sodium intake causes the body to retain water and artificially increases blood volume. The heart, already swollen and tired from the blood leaking backward, has to make more effort and work against a higher pressure to pump this extra water load increased within the vessels. Likewise, saturated fats, processed meat products, and excessive sugar consumption harden vessel walls, cause high blood pressure, and lead to long-term blockage of the heart vessels, seriously worsening the patient’s overall cardiovascular risk profile.
Why Are Interventional Treatments Increasingly Becoming the First Choice in Modern Medicine for Aortic Valve Insufficiency?
The technological revolutions experienced in the field of cardiology over the last decade have fundamentally changed the philosophy of approaching heart diseases. Our aim is not only to keep the patient alive, but also to provide the highest quality of life, with the lowest risk of complications and in the fastest way possible. These incision-free, restriction-free, and much less traumatic options offered by interventional methods provide patients with great psychological confidence and relief as well.
Today, when planning a patient’s treatment, it is no longer the decision of a single doctor but the shared wisdom of heart teams in which interventional cardiologists, cardiac surgeons, imaging specialists, and anesthesiologists sit at the same table. Each patient’s vascular structure, heart functions, and general health status are analyzed one by one. Considering the extraordinary clinical success rates and recovery speeds achieved, innovative valve repairs performed through the groin have gone far beyond being merely a necessity or an alternative.

Prof. Dr. Kadriye Orta Kılıçkesmez is one of the leading figures in the field of Turkish cardiology. She was born on January 24, 1974, in Tekirdağ. After completing her undergraduate education at Istanbul University Cerrahpaşa Faculty of Medicine, she chose cardiology as her specialty and received her specialist training at the Cardiology Institute of the same university. In 2015, she was appointed by the university to establish the Şişli Etfal cardiology clinic and Angio laboratory. Becoming a professor in 2017, Kadriye Kılıçkesmez established the cardiology clinic and Angio laboratory of Prof. Dr. Cemil Taşçı Hospital in 2020 and ensured that the clinic became a training clinic.